
- •Contents
- •Preface to the first edition
- •Flagella
- •Cell walls and mucilages
- •Plastids
- •Mitochondria and peroxisomes
- •Division of chloroplasts and mitochondria
- •Storage products
- •Contractile vacuoles
- •Nutrition
- •Gene sequencing and algal systematics
- •Classification
- •Algae and the fossil record
- •REFERENCES
- •CYANOPHYCEAE
- •Morphology
- •Cell wall and gliding
- •Pili and twitching
- •Sheaths
- •Protoplasmic structure
- •Gas vacuoles
- •Pigments and photosynthesis
- •Akinetes
- •Heterocysts
- •Nitrogen fixation
- •Asexual reproduction
- •Growth and metabolism
- •Lack of feedback control of enzyme biosynthesis
- •Symbiosis
- •Extracellular associations
- •Ecology of cyanobacteria
- •Freshwater environment
- •Terrestrial environment
- •Adaption to silting and salinity
- •Cyanotoxins
- •Cyanobacteria and the quality of drinking water
- •Utilization of cyanobacteria as food
- •Cyanophages
- •Secretion of antibiotics and siderophores
- •Calcium carbonate deposition and fossil record
- •Chroococcales
- •Classification
- •Oscillatoriales
- •Nostocales
- •REFERENCES
- •REFERENCES
- •REFERENCES
- •RHODOPHYCEAE
- •Cell structure
- •Cell walls
- •Chloroplasts and storage products
- •Pit connections
- •Calcification
- •Secretory cells
- •Iridescence
- •Epiphytes and parasites
- •Defense mechanisms of the red algae
- •Commercial utilization of red algal mucilages
- •Reproductive structures
- •Carpogonium
- •Spermatium
- •Fertilization
- •Meiosporangia and meiospores
- •Asexual spores
- •Spore motility
- •Classification
- •Cyanidiales
- •Porphyridiales
- •Bangiales
- •Acrochaetiales
- •Batrachospermales
- •Nemaliales
- •Corallinales
- •Gelidiales
- •Gracilariales
- •Ceramiales
- •REFERENCES
- •Cell structure
- •Phototaxis and eyespots
- •Asexual reproduction
- •Sexual reproduction
- •Classification
- •Position of flagella in cells
- •Flagellar roots
- •Multilayered structure
- •Occurrence of scales or a wall on the motile cells
- •Cell division
- •Superoxide dismutase
- •Prasinophyceae
- •Charophyceae
- •Classification
- •Klebsormidiales
- •Zygnematales
- •Coleochaetales
- •Charales
- •Ulvophyceae
- •Classification
- •Ulotrichales
- •Ulvales
- •Cladophorales
- •Dasycladales
- •Caulerpales
- •Siphonocladales
- •Chlorophyceae
- •Classification
- •Volvocales
- •Tetrasporales
- •Prasiolales
- •Chlorellales
- •Trebouxiales
- •Sphaeropleales
- •Chlorosarcinales
- •Chaetophorales
- •Oedogoniales
- •REFERENCES
- •REFERENCES
- •EUGLENOPHYCEAE
- •Nucleus and nuclear division
- •Eyespot, paraflagellar swelling, and phototaxis
- •Muciferous bodies and extracellular structures
- •Chloroplasts and storage products
- •Nutrition
- •Classification
- •Heteronematales
- •Eutreptiales
- •Euglenales
- •REFERENCES
- •DINOPHYCEAE
- •Cell structure
- •Theca
- •Scales
- •Flagella
- •Pusule
- •Chloroplasts and pigments
- •Phototaxis and eyespots
- •Nucleus
- •Projectiles
- •Accumulation body
- •Resting spores or cysts or hypnospores and fossil Dinophyceae
- •Toxins
- •Dinoflagellates and oil and coal deposits
- •Bioluminescence
- •Rhythms
- •Heterotrophic dinoflagellates
- •Direct engulfment of prey
- •Peduncle feeding
- •Symbiotic dinoflagellates
- •Classification
- •Prorocentrales
- •Dinophysiales
- •Peridiniales
- •Gymnodiniales
- •REFERENCES
- •REFERENCES
- •Chlorarachniophyta
- •REFERENCES
- •CRYPTOPHYCEAE
- •Cell structure
- •Ecology
- •Symbiotic associations
- •Classification
- •Goniomonadales
- •Cryptomonadales
- •Chroomonadales
- •REFERENCES
- •CHRYSOPHYCEAE
- •Cell structure
- •Flagella and eyespot
- •Internal organelles
- •Extracellular deposits
- •Statospores
- •Nutrition
- •Ecology
- •Classification
- •Chromulinales
- •Parmales
- •Chrysomeridales
- •REFERENCES
- •SYNUROPHYCEAE
- •Classification
- •REFERENCES
- •EUSTIGMATOPHYCEAE
- •REFERENCES
- •PINGUIOPHYCEAE
- •REFERENCES
- •DICTYOCHOPHYCEAE
- •Classification
- •Rhizochromulinales
- •Pedinellales
- •Dictyocales
- •REFERENCES
- •PELAGOPHYCEAE
- •REFERENCES
- •BOLIDOPHYCEAE
- •REFERENCE
- •BACILLARIOPHYCEAE
- •Cell structure
- •Cell wall
- •Cell division and the formation of the new wall
- •Extracellular mucilage, biolfouling, and gliding
- •Motility
- •Plastids and storage products
- •Resting spores and resting cells
- •Auxospores
- •Rhythmic phenomena
- •Physiology
- •Chemical defense against predation
- •Ecology
- •Marine environment
- •Freshwater environment
- •Fossil diatoms
- •Classification
- •Biddulphiales
- •Bacillariales
- •REFERENCES
- •RAPHIDOPHYCEAE
- •REFERENCES
- •XANTHOPHYCEAE
- •Cell structure
- •Cell wall
- •Chloroplasts and food reserves
- •Asexual reproduction
- •Sexual reproduction
- •Mischococcales
- •Tribonematales
- •Botrydiales
- •Vaucheriales
- •REFERENCES
- •PHAEOTHAMNIOPHYCEAE
- •REFERENCES
- •PHAEOPHYCEAE
- •Cell structure
- •Cell walls
- •Flagella and eyespot
- •Chloroplasts and photosynthesis
- •Phlorotannins and physodes
- •Life history
- •Classification
- •Dictyotales
- •Sphacelariales
- •Cutleriales
- •Desmarestiales
- •Ectocarpales
- •Laminariales
- •Fucales
- •REFERENCES
- •PRYMNESIOPHYCEAE
- •Cell structure
- •Flagella
- •Haptonema
- •Chloroplasts
- •Other cytoplasmic structures
- •Scales and coccoliths
- •Toxins
- •Classification
- •Prymnesiales
- •Pavlovales
- •REFERENCES
- •Toxic algae
- •Toxic algae and the end-Permian extinction
- •Cooling of the Earth, cloud condensation nuclei, and DMSP
- •Chemical defense mechanisms of algae
- •The Antarctic and Southern Ocean
- •The grand experiment
- •Antarctic lakes as a model for life on the planet Mars or Jupiter’s moon Europa
- •Ultraviolet radiation, the ozone hole, and sunscreens produced by algae
- •Hydrogen fuel cells and hydrogen gas production by algae
- •REFERENCES
- •Glossary
- •Index
Chapter 19
Heterokontophyta
XANTHOPHYCEAE
The Xanthophyceae contain primarily freshwater and terrestrial algae with a few marine representatives. The class is characterized by motile cells with a forwardly directed tinsel flagellum and a posteriorly directed whiplash flagellum (Figs. 19.1, 19.5(c)). The chloroplasts contain chlorophylls a and c (Sullivan et al., 1990), lack fucoxanthin, and are colored yellowish-green. The eyespot in motile cells is always in the chloroplast (Figs. 19.1, 19.5(c)), and the chloroplasts are surrounded by two membranes of chloroplast endoplasmic reticulum. The outer membrane of the chloroplast E.R. is usually continuous with the outer membrane of the nucleus. In most non-motile cells the wall is composed of two overlapping halves (Figs. 19.2 (d), (e), (f), 19.3, 19.4). Molecular data have shown the Xanthophyceae is most closely related to the Phaeophyceae (Ariztia et al., 1991; Potter et al., 1997). Although the class is commonly called the Xanthophyceae, the proper name is the Tribophyceae since there is no genus in the class that can lend its name to Xanthophyceae (Hibberd, 1981).
Cell structure
Cell wall
The cell walls of two Xanthophyceae, Tribonema (Figs. 19.2, 19.3) (Cleare and Percival, 1973) and Vaucheria (Figs. 19.7, 19.8), are composed of cellulose (Parker et al., 1963). In Vaucheria cellulose comprises 90% of the wall, with the remaining
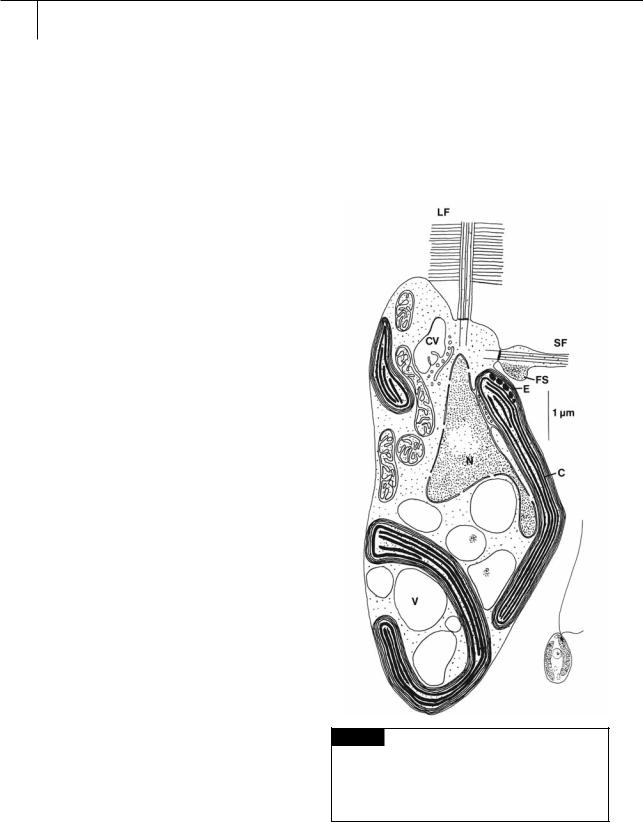
Fig. 19.1 Light and electron microscopical drawing of a zoospore of a typical member of the Xanthophyceae,
Mischococcus sphaerocephalus. (C) Chloroplast; (CV) contractile vacuole; (E) eyespot; (FS) flagellar swelling; (LF) long flagellum with hairs; (N) nucleus; (SF) short flagellum;
(V) vacuole. (Adapted from Hibberd and Leedale, 1971.)

414 CHLOROPLAST E.R.: EVOLUTION OF TWO MEMBRANES
Fig. 19.2 Light microscopy of filaments of Tribonema regulare (a) and Tribonema utriculosum (b). (c)
Tribonema viride, zoosporangium containing two zoospores. (d) Liberated zoospores of Tribonema regulare. (e) H-shaped cell walls of
Tribonema regulare remaining after liberation of zoospores. (f) Scanning electron micrograph of a two-celled filament of Tribonema viridae showing the margin of an overlapping H-piece (arrows). (From Lokhorst and Star, 2003.)
Fig. 19.3 Wall structure of
Tribonema bombycinum, after treatment with potassium hydroxide. (a) Two H-pieces articulated to enclose a single protoplast. (b), (c) Recently divided cell showing the intercalation of a new H-piece. (After Smith, 1938.)
portion being amorphous polysaccharides composed primarily of glucose and uronic acids.
Many of the algae in the class have walls composed of two overlapping halves that fit together as do the two parts of the bacteriologist’s Petri dish (Figs. 19.2(d), (e), (f), 19.3, 19.4). The two-part nature of the wall cannot be delineated with the light microscope unless the cells have been treated with certain reagents such as concentrated potassium hydroxide. A typical example is the wall of Ophiocytium majus (one study places Ophiocytium in the closely related Eustigmatophyceae based on the sequence of
mitochondrial cytochrome oxidase (Ehara et al., 1997)), which is tubular in shape (Fig. 19.4). The wall is composed of two parts, a cap of constant size fitted over a tubular basal portion. As the cell grows and increases in length, the tubular basal portion elongates, but the cap remains the same size. The rims of both the cap and the tube are finely tapered and overlap for some distance so that the bipartite condition is not normally apparent in living cells. A layer of intercalary material, presumably functioning as a cementing substance, separates the cap from the tubular part of the wall. Filamentous genera, such as
