
- •Contents
- •Preface to the first edition
- •Flagella
- •Cell walls and mucilages
- •Plastids
- •Mitochondria and peroxisomes
- •Division of chloroplasts and mitochondria
- •Storage products
- •Contractile vacuoles
- •Nutrition
- •Gene sequencing and algal systematics
- •Classification
- •Algae and the fossil record
- •REFERENCES
- •CYANOPHYCEAE
- •Morphology
- •Cell wall and gliding
- •Pili and twitching
- •Sheaths
- •Protoplasmic structure
- •Gas vacuoles
- •Pigments and photosynthesis
- •Akinetes
- •Heterocysts
- •Nitrogen fixation
- •Asexual reproduction
- •Growth and metabolism
- •Lack of feedback control of enzyme biosynthesis
- •Symbiosis
- •Extracellular associations
- •Ecology of cyanobacteria
- •Freshwater environment
- •Terrestrial environment
- •Adaption to silting and salinity
- •Cyanotoxins
- •Cyanobacteria and the quality of drinking water
- •Utilization of cyanobacteria as food
- •Cyanophages
- •Secretion of antibiotics and siderophores
- •Calcium carbonate deposition and fossil record
- •Chroococcales
- •Classification
- •Oscillatoriales
- •Nostocales
- •REFERENCES
- •REFERENCES
- •REFERENCES
- •RHODOPHYCEAE
- •Cell structure
- •Cell walls
- •Chloroplasts and storage products
- •Pit connections
- •Calcification
- •Secretory cells
- •Iridescence
- •Epiphytes and parasites
- •Defense mechanisms of the red algae
- •Commercial utilization of red algal mucilages
- •Reproductive structures
- •Carpogonium
- •Spermatium
- •Fertilization
- •Meiosporangia and meiospores
- •Asexual spores
- •Spore motility
- •Classification
- •Cyanidiales
- •Porphyridiales
- •Bangiales
- •Acrochaetiales
- •Batrachospermales
- •Nemaliales
- •Corallinales
- •Gelidiales
- •Gracilariales
- •Ceramiales
- •REFERENCES
- •Cell structure
- •Phototaxis and eyespots
- •Asexual reproduction
- •Sexual reproduction
- •Classification
- •Position of flagella in cells
- •Flagellar roots
- •Multilayered structure
- •Occurrence of scales or a wall on the motile cells
- •Cell division
- •Superoxide dismutase
- •Prasinophyceae
- •Charophyceae
- •Classification
- •Klebsormidiales
- •Zygnematales
- •Coleochaetales
- •Charales
- •Ulvophyceae
- •Classification
- •Ulotrichales
- •Ulvales
- •Cladophorales
- •Dasycladales
- •Caulerpales
- •Siphonocladales
- •Chlorophyceae
- •Classification
- •Volvocales
- •Tetrasporales
- •Prasiolales
- •Chlorellales
- •Trebouxiales
- •Sphaeropleales
- •Chlorosarcinales
- •Chaetophorales
- •Oedogoniales
- •REFERENCES
- •REFERENCES
- •EUGLENOPHYCEAE
- •Nucleus and nuclear division
- •Eyespot, paraflagellar swelling, and phototaxis
- •Muciferous bodies and extracellular structures
- •Chloroplasts and storage products
- •Nutrition
- •Classification
- •Heteronematales
- •Eutreptiales
- •Euglenales
- •REFERENCES
- •DINOPHYCEAE
- •Cell structure
- •Theca
- •Scales
- •Flagella
- •Pusule
- •Chloroplasts and pigments
- •Phototaxis and eyespots
- •Nucleus
- •Projectiles
- •Accumulation body
- •Resting spores or cysts or hypnospores and fossil Dinophyceae
- •Toxins
- •Dinoflagellates and oil and coal deposits
- •Bioluminescence
- •Rhythms
- •Heterotrophic dinoflagellates
- •Direct engulfment of prey
- •Peduncle feeding
- •Symbiotic dinoflagellates
- •Classification
- •Prorocentrales
- •Dinophysiales
- •Peridiniales
- •Gymnodiniales
- •REFERENCES
- •REFERENCES
- •Chlorarachniophyta
- •REFERENCES
- •CRYPTOPHYCEAE
- •Cell structure
- •Ecology
- •Symbiotic associations
- •Classification
- •Goniomonadales
- •Cryptomonadales
- •Chroomonadales
- •REFERENCES
- •CHRYSOPHYCEAE
- •Cell structure
- •Flagella and eyespot
- •Internal organelles
- •Extracellular deposits
- •Statospores
- •Nutrition
- •Ecology
- •Classification
- •Chromulinales
- •Parmales
- •Chrysomeridales
- •REFERENCES
- •SYNUROPHYCEAE
- •Classification
- •REFERENCES
- •EUSTIGMATOPHYCEAE
- •REFERENCES
- •PINGUIOPHYCEAE
- •REFERENCES
- •DICTYOCHOPHYCEAE
- •Classification
- •Rhizochromulinales
- •Pedinellales
- •Dictyocales
- •REFERENCES
- •PELAGOPHYCEAE
- •REFERENCES
- •BOLIDOPHYCEAE
- •REFERENCE
- •BACILLARIOPHYCEAE
- •Cell structure
- •Cell wall
- •Cell division and the formation of the new wall
- •Extracellular mucilage, biolfouling, and gliding
- •Motility
- •Plastids and storage products
- •Resting spores and resting cells
- •Auxospores
- •Rhythmic phenomena
- •Physiology
- •Chemical defense against predation
- •Ecology
- •Marine environment
- •Freshwater environment
- •Fossil diatoms
- •Classification
- •Biddulphiales
- •Bacillariales
- •REFERENCES
- •RAPHIDOPHYCEAE
- •REFERENCES
- •XANTHOPHYCEAE
- •Cell structure
- •Cell wall
- •Chloroplasts and food reserves
- •Asexual reproduction
- •Sexual reproduction
- •Mischococcales
- •Tribonematales
- •Botrydiales
- •Vaucheriales
- •REFERENCES
- •PHAEOTHAMNIOPHYCEAE
- •REFERENCES
- •PHAEOPHYCEAE
- •Cell structure
- •Cell walls
- •Flagella and eyespot
- •Chloroplasts and photosynthesis
- •Phlorotannins and physodes
- •Life history
- •Classification
- •Dictyotales
- •Sphacelariales
- •Cutleriales
- •Desmarestiales
- •Ectocarpales
- •Laminariales
- •Fucales
- •REFERENCES
- •PRYMNESIOPHYCEAE
- •Cell structure
- •Flagella
- •Haptonema
- •Chloroplasts
- •Other cytoplasmic structures
- •Scales and coccoliths
- •Toxins
- •Classification
- •Prymnesiales
- •Pavlovales
- •REFERENCES
- •Toxic algae
- •Toxic algae and the end-Permian extinction
- •Cooling of the Earth, cloud condensation nuclei, and DMSP
- •Chemical defense mechanisms of algae
- •The Antarctic and Southern Ocean
- •The grand experiment
- •Antarctic lakes as a model for life on the planet Mars or Jupiter’s moon Europa
- •Ultraviolet radiation, the ozone hole, and sunscreens produced by algae
- •Hydrogen fuel cells and hydrogen gas production by algae
- •REFERENCES
- •Glossary
- •Index
CYANOBACTERIA 49
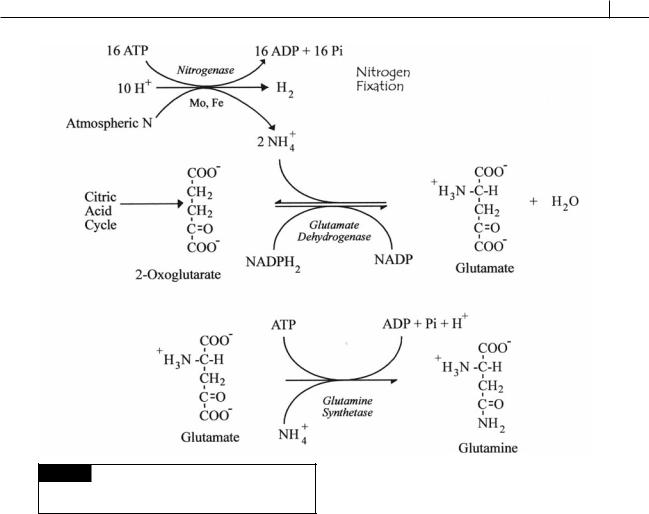
Fig. 2.27 The chemistry of nitrogen fixation and the
subsequent incorporation of the fixed nitrogen into
glutamate and glutamine.
have a limited period of physiological activity and appear to have a limited life. Senescent heterocysts undergo vacuolation and usually break off from the filament, causing fragmentation of the filament.
Heterocysts are dependent on a supply of substrates from adjacent vegetative cells through cytoplasmic connections (microplasmodesmata). These cytoplasmic connections probably convey nitrogen fixed in the form of glutamine (Fig. 2.27) by the heterocysts to vegetative cells. The vegetative cells transfer photosynthate to the heterocysts since the heterocysts are incapable of carbon fixation.
Nitrogen fixation
Cyanobacteria are diazotrophs (able to fix atmospheric nitrogen). All known nitrogen-fixing organ-
isms are prokaryotes. In nitrogen fixation, N2 from the atmosphere is fixed by the enzyme nitrogenase into ammonium using ATP as a source of energy (Fig. 2.27). The process is one of the most metabolically expensive processes in biology, requiring 16 ATP for each molecule of N2 fixed. The amount of biologically fixed nitrogen produced is in excess of 2 1013 g year 1. In contrast, lightning discharge, the primary abiotic source of fixed nitrogen, accounts for 5 1012 g year 1 (Raymond et al., 2004).
The ammonium fixed by nitrogen fixation is added to 2-oxoglutarate (from the citric acid cycle) by the enzyme glutamate dehydrogenase to form glutamate (glutamic acid) (Fig. 2.27). Addition of a second ammonium to glutamate produces glutamine, the molecule that is transferred from one cyanobacterial cell to another.
In bacteria, nitrogenase is composed of two components, dinitrogenase reductase (iron protein) and dinitrogenase (molybdenum-iron protein) encoded by the nif HDK operon (Henson et al.,
50 THE PROKARYOTIC ALGAE
2004). The situation is probably similar in the cyanobacteria.
Hydrogen gas is also produced in nitrogen fixation (Fig. 2.27) and has drawn interest as a renewable energy source as hydrogen fuel-cell technology for motor vehicles becomes more of a practical reality (Schutz et al., 2004).
Nitrogenase, the nitrogen-fixing enzyme, is very sensitive to inactivation by oxygen. Cyanobacteria have evolved three different mechanisms designed to exclude oxygen from the area of the cells containing nitrogenase:
1Heterocystous cyanobacteria: These cyanobacteria occur primarily in fresh and brackish water and fix nitrogen in heterocysts. Heterocysts are surrounded by a glycolipid layer which is
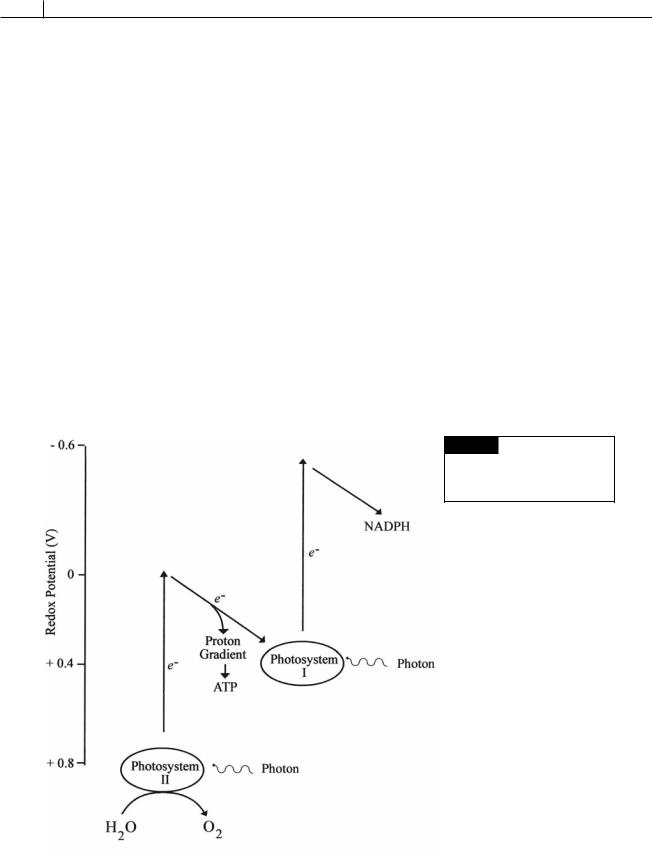
impermeable to O2 (Staal et al., 2003). Heterocysts lack photosystem II (Fig. 2.28) and,
therefore, the ability to evolve O2. Heterocysts do have cyclic photophosphorulation (Fig.
2.29) and can produce the ATP necessary for
nitrogen fixation. Heterocysts also have a form of myoglobin called cyanoglobin that scavenges oxygen, preventing inhibition of nitrogenase (Potts et al., 1992). Under anaerobic conditions, in an atmosphere of nitrogen and carbon dioxide, both vegetative cells and heterocysts can fix nitrogen.
2Non-filamentous cyanobacteria that fix nitrogen in the dark but not in the light: These cyanobacteria fix nitrogen in the dark when photosynthesis is not producing nitrogenase-inhibiting oxygen. If Synechococcus is grown under a 12hour light: 12-hour dark cycle, most of the nitrogen fixation occurs during the dark period (Fig. 2.30). If the cells are subjected
to continuous illumination, an endogenous timing cycle entrained by cell division continues to alternate the level of photosynthesis and nitrogen fixation (Mitsui et al., 1986; Chen et al., 1996). Nitrogenase activity peaks at the same time that it had in the previous dark period
Fig. 2.28 Non-cyclic
photophosphorulation. Heterocysts
in cyanobacteria lack photosystem II
and do not produce oxygen.
CYANOBACTERIA 51
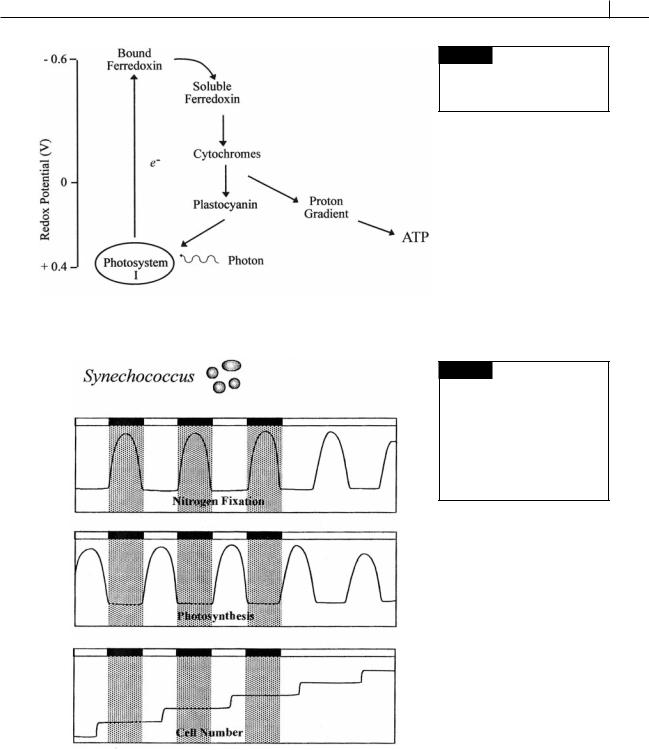
Fig. 2.29 Cyclic photophosphorulation. Heterocysts in cyanobacteria produce ATP by cyclic photophosphorulation.
Fig. 2.30 Illustration of the relationship between photosynthesis and nitrogen fixation in a culture of
Synechococcus sp. growing under a 12-hour light: 12-hour dark photoperiod. Under continuous illumination, an endogenous clock maintains the cycles. (After Mitsui et al., 1986.)
(Fig. 2.30). At the beginning of the next light |
beginning of the next dark period for nitrogen |
period, oxygen is produced by photosynthesis |
fixation to occur. |
and the nitrogenase is inactivated. New |
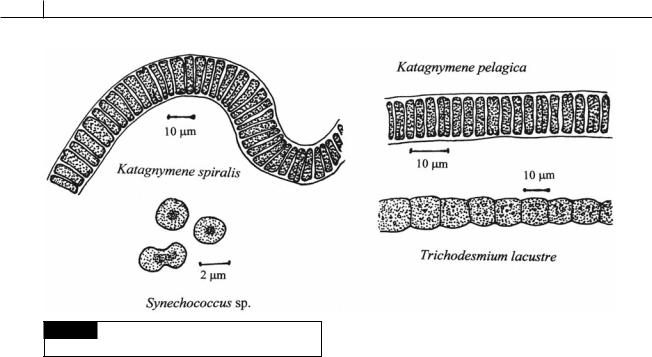
3 Trichodesmium (Figs. 2.31, 2.56(g)) and |
nitrogenase must be synthesized at the |
Katagnymene (Fig. 2.31): These cyanobacteria |
52 THE PROKARYOTIC ALGAE
Fig. 2.31 Some nitrogen-fixing cyanobacteria that lack
heterocysts.
are the major bloom-forming, nitrogen-fixing, organisms in the oceans, responsible for fixing one-quarter of the total nitrogen in the oceans of the world (Bergman and Carpenter, 1991).
These filamentous cyanobacteria do not have heterocysts yet fix nitrogen in the light under aerobic conditions (Bergman et al., 1997).
Within the filaments, 10 to 15% of the cells (called diazocytes) are specialized to fix nitrogen, while the others do not (Lundgren et al., 2001). Cells that fix nitrogen are adjacent to one another and have a denser thylakoid network with fewer gas vacuoles and cyanophycin granules (Fredriksson and Bergman, 1997). The tropical seas where
Trichodesmium and Katagnymene live are relatively low in dissolved oxygen and this may assist the nitrogen-fixing cells in maintaining anaerobic conditions in the protoplasm where nitrogenase is present (Staal, Meysman and Stal, 2003).
Trichodesmium and Katagnymene
represent the most ancient type of nitrogenfixing cyanobacteria (Berman-Frank et al., 2001).
Circadian rhythms
The cyanobacteria have circadian rhythms in photosynthesis, nitrogen fixation, and cell division similar to those in eukaryotic organisms (Fig. 2.30). The requirements of a circadian rhythm are: (1) approximate 24-hour cycles in biological processes even in the absence of an environmental cycle; (2) synchronization with the environment through light or environmental cues; and (3) maintenance of a nearly constant period over a range of physiologically relevant temperatures (Golden, 2003).
The details of the cyanobacterial circadian rhythm have been elucidated in Synechococcus elongatus (Williams et al., 2002). The key components of the timekeeping complex are the proteins KaiA, KaiB, and KaiC coded by the genes KaiA, KaiB, and KaiC (Figs. 2.32, 2.33). Interactions between these three proteins maintain the rhythm at about 24 hours. In the real world, the clock is reset daily by cycles of light and darkness, temperature and humidity. The major portal into the clock is the protein kinase CikA. Environmental change to CikA changes the configuration of the protein KaiA which causes phosphorulation of the protein KaiC (Fig. 2.32), resetting the clock. The output that sets cycles of nitrogen fixation, photosynthesis, etc. begins when KaiC interacts with the protein SasA (Synechococcus adaptive sensor). This results in information being relayed downstream to produce the observed effect.
