
- •Contents
- •Contributors
- •1 Introduction
- •2.1 Posterior Compartment
- •2.2 Anterior Compartment
- •2.3 Middle Compartment
- •2.4 Perineal Body
- •3 Compartments
- •3.1 Posterior Compartment
- •3.1.1 Connective Tissue Structures
- •3.1.2 Muscles
- •3.1.3 Reinterpreted Anatomy and Clinical Relevance
- •3.2 Anterior Compartment
- •3.2.1 Connective Tissue Structures
- •3.2.2 Muscles
- •3.2.3 Reinterpreted Anatomy and Clinical Relevance
- •3.2.4 Important Vessels, Nerves, and Lymphatics of the Anterior Compartment
- •3.3 Middle Compartment
- •3.3.1 Connective Tissue Structures
- •3.3.2 Muscles
- •3.3.3 Reinterpreted Anatomy and Clinical Relevance
- •3.3.4 Important Vessels, Nerves, and Lymphatics of the Middle Compartment
- •4 Perineal Body
- •References
- •MR and CT Techniques
- •1 Introduction
- •2.1 Introduction
- •2.2.1 Spasmolytic Medication
- •2.3.2 Diffusion-Weighted Imaging
- •2.3.3 Dynamic Contrast Enhancement
- •3 CT Technique
- •3.1 Introduction
- •3.2 Technical Disadvantages
- •3.4 Oral and Rectal Contrast
- •References
- •Uterus: Normal Findings
- •1 Introduction
- •References
- •1 Clinical Background
- •1.1 Epidemiology
- •1.2 Clinical Presentation
- •1.3 Embryology
- •1.4 Pathology
- •2 Imaging
- •2.1 Technique
- •2.2.1 Class I Anomalies: Dysgenesis
- •2.2.2 Class II Anomalies: Unicornuate Uterus
- •2.2.3 Class III Anomalies: Uterus Didelphys
- •2.2.4 Class IV Anomalies: Bicornuate Uterus
- •2.2.5 Class V Anomalies: Septate Uterus
- •2.2.6 Class VI Anomalies: Arcuate Uterus
- •2.2.7 Class VII Anomalies
- •References
- •Benign Uterine Lesions
- •1 Background
- •1.1 Uterine Leiomyomas
- •1.1.1 Epidemiology
- •1.1.2 Pathogenesis
- •1.1.3 Histopathology
- •1.1.4 Clinical Presentation
- •1.1.5 Therapy
- •1.1.5.1 Indications
- •1.1.5.2 Medical Therapy and Ablation
- •1.1.5.3 Surgical Therapy
- •1.1.5.4 Uterine Artery Embolization (UAE)
- •1.1.5.5 Magnetic Resonance-Guided Focused Ultrasound
- •2 Adenomyosis of the Uterus
- •2.1 Epidemiology
- •2.2 Pathogenesis
- •2.3 Histopathology
- •2.4 Clinical Presentation
- •2.5 Therapy
- •3 Imaging
- •3.2 Magnetic Resonance Imaging
- •3.2.1 Magnetic Resonance Imaging: Technique
- •3.2.2 MR Appearance of Uterine Leiomyomas
- •3.2.3 Locations, Growth Patterns, and Imaging Characteristics
- •3.2.4 Histologic Subtypes and Forms of Degeneration
- •3.2.5 Differential Diagnosis
- •3.2.6 MR Appearance of Uterine Adenomyosis
- •3.2.7 Locations, Growth Patterns, and Imaging Characteristics
- •3.2.8 Differential Diagnosis
- •3.3 Computed Tomography
- •3.3.1 CT Technique
- •3.3.2 CT Appearance of Uterine Leiomyoma and Adenomyosis
- •3.3.3 Atypical Appearances on CT and Differential Diagnosis
- •4.1 Indications
- •4.2 Technique
- •Bibliography
- •Cervical Cancer
- •1 Background
- •1.1 Epidemiology
- •1.2 Pathogenesis
- •1.3 Screening
- •1.4 HPV Vaccination
- •1.5 Clinical Presentation
- •1.6 Histopathology
- •1.7 Staging
- •1.8 Growth Patterns
- •1.9 Treatment
- •1.9.1 Treatment of Microinvasive Cervical Cancer
- •1.9.2 Treatment of Grossly Invasive Cervical Carcinoma (FIGO IB-IVA)
- •1.9.3 Treatment of Recurrent Disease
- •1.9.4 Treatment of Cervical Cancer During Pregnancy
- •1.10 Prognosis
- •2 Imaging
- •2.1 Indications
- •2.1.1 Role of CT and MRI
- •2.2 Imaging Technique
- •2.2.2 Dynamic MRI
- •2.2.3 Coil Technique
- •2.2.4 Vaginal Opacification
- •2.3 Staging
- •2.3.1 General MR Appearance
- •2.3.2 Rare Histologic Types
- •2.3.3 Tumor Size
- •2.3.4 Local Staging
- •2.3.4.1 Stage IA
- •2.3.4.2 Stage IB
- •2.3.4.3 Stage IIA
- •2.3.4.4 Stage IIB
- •2.3.4.5 Stage IIIA
- •2.3.4.6 Stage IIIB
- •2.3.4.7 Stage IVA
- •2.3.4.8 Stage IVB
- •2.3.5 Lymph Node Staging
- •2.3.6 Distant Metastases
- •2.4 Specific Diagnostic Queries
- •2.4.1 Preoperative Imaging
- •2.4.2 Imaging Before Radiotherapy
- •2.5 Follow-Up
- •2.5.1 Findings After Surgery
- •2.5.2 Findings After Chemotherapy
- •2.5.3 Findings After Radiotherapy
- •2.5.4 Recurrent Cervical Cancer
- •2.6.1 Ultrasound
- •2.7.1 Metastasis
- •2.7.2 Malignant Melanoma
- •2.7.3 Lymphoma
- •2.8 Benign Lesions of the Cervix
- •2.8.1 Nabothian Cyst
- •2.8.2 Leiomyoma
- •2.8.3 Polyps
- •2.8.4 Rare Benign Tumors
- •2.8.5 Cervicitis
- •2.8.6 Endometriosis
- •2.8.7 Ectopic Cervical Pregnancy
- •References
- •Endometrial Cancer
- •1.1 Epidemiology
- •1.2 Pathology and Risk Factors
- •1.3 Symptoms and Diagnosis
- •2 Endometrial Cancer Staging
- •2.1 MR Protocol for Staging Endometrial Carcinoma
- •2.2.1 Stage I Disease
- •2.2.2 Stage II Disease
- •2.2.3 Stage III Disease
- •2.2.4 Stage IV Disease
- •4 Therapeutic Approaches
- •4.1 Surgery
- •4.2 Adjuvant Treatment
- •4.3 Fertility-Sparing Treatment
- •5.1 Treatment of Recurrence
- •6 Prognosis
- •References
- •Uterine Sarcomas
- •1 Epidemiology
- •2 Pathology
- •2.1 Smooth Muscle Tumours
- •2.2 Endometrial Stromal Tumours
- •3 Clinical Background
- •4 Staging
- •5 Imaging
- •5.1 Leiomyosarcoma
- •5.2.3 Undifferentiated Uterine Sarcoma
- •5.3 Adenosarcoma
- •6 Prognosis and Treatment
- •References
- •1.1 Anatomical Relationships
- •1.4 Pelvic Fluid
- •2 Developmental Anomalies
- •2.1 Congenital Abnormalities
- •2.2 Ovarian Maldescent
- •3 Ovarian Transposition
- •References
- •1 Introduction
- •4 Benign Adnexal Lesions
- •4.1.1 Physiological Ovarian Cysts: Follicular and Corpus Luteum Cysts
- •4.1.1.1 Imaging Findings in Physiological Ovarian Cysts
- •4.1.1.2 Differential Diagnosis
- •4.1.2 Paraovarian Cysts
- •4.1.2.1 Imaging Findings
- •4.1.2.2 Differential Diagnosis
- •4.1.3 Peritoneal Inclusion Cysts
- •4.1.3.1 Imaging Findings
- •4.1.3.2 Differential Diagnosis
- •4.1.4 Theca Lutein Cysts
- •4.1.4.1 Imaging Findings
- •4.1.4.2 Differential Diagnosis
- •4.1.5 Polycystic Ovary Syndrome
- •4.1.5.1 Imaging Findings
- •4.1.5.2 Differential Diagnosis
- •4.2.1 Cystadenoma
- •4.2.1.1 Imaging Findings
- •4.2.1.2 Differential Diagnosis
- •4.2.2 Cystadenofibroma
- •4.2.2.1 Imaging Features
- •4.2.3 Mature Teratoma
- •4.2.3.1 Mature Cystic Teratoma
- •Imaging Findings
- •Differential Diagnosis
- •4.2.3.2 Monodermal Teratoma
- •Imaging Findings
- •4.2.4 Benign Sex Cord-Stromal Tumors
- •4.2.4.1 Fibroma and Thecoma
- •Imaging Findings
- •4.2.4.2 Sclerosing Stromal Tumor
- •Imaging Findings
- •4.2.5 Brenner Tumors
- •4.2.5.1 Imaging Findings
- •4.2.5.2 Differential Diagnosis
- •5 Functioning Ovarian Tumors
- •References
- •1 Introduction
- •2.1 Context
- •2.2.2 Indications According to Simple Rules
- •References
- •CT and MRI in Ovarian Carcinoma
- •1 Introduction
- •2.1 Familial or Hereditary Ovarian Cancers
- •3 Screening for Ovarian Cancer
- •5 Tumor Markers
- •6 Clinical Presentation
- •7 Imaging of Ovarian Cancer
- •7.1.2 Peritoneal Carcinomatosis
- •7.1.3 Ascites
- •7.3 Staging of Ovarian Cancer
- •7.3.1 Staging by CT and MRI
- •Imaging Findings According to Tumor Stages
- •Value of Imaging
- •7.3.2 Prediction of Resectability
- •7.4 Tumor Types
- •7.4.1 Epithelial Ovarian Cancer
- •High-Grade Serous Ovarian Cancer
- •Low-Grade Serous Ovarian Cancer
- •Mucinous Epithelial Ovarian Cancer
- •Endometrioid Ovarian Carcinomas
- •Clear Cell Carcinomas
- •Imaging Findings of Epithelial Ovarian Cancers
- •Differential Diagnosis
- •Borderline Tumors
- •Imaging Findings
- •Differential Diagnosis
- •Recurrent Ovarian Cancer
- •Imaging Findings
- •Differential Diagnosis
- •Value of Imaging
- •Malignant Germ Cell Tumors
- •Dysgerminomas
- •Imaging Findings
- •Differential Diagnosis
- •Immature Teratomas
- •Imaging Findings
- •Malignant Transformation in Benign Teratoma
- •Imaging Findings
- •Differential Diagnosis
- •Sex-Cord Stromal Tumors
- •Granulosa Cell Tumors
- •Imaging Findings
- •Sertoli-Leydig Cell Tumor
- •Imaging Findings
- •Ovarian Lymphoma
- •Imaging Findings
- •Differential Diagnosis
- •7.4.3 Ovarian Metastases
- •Imaging Findings
- •Differential Diagnosis
- •7.5 Fallopian Tube Cancer
- •7.5.1 Imaging Findings
- •Differential Diagnosis
- •References
- •Endometriosis
- •1 Introduction
- •2.1 Sonography
- •3 MR Imaging Findings
- •References
- •Vagina and Vulva
- •1 Introduction
- •3.1 CT Appearance
- •3.2 MRI Protocol
- •3.3 MRI Appearance
- •4.1 Imperforate Hymen
- •4.2 Congenital Vaginal Septa
- •4.3 Vaginal Agenesis
- •5.1 Vaginal Cysts
- •5.1.1 Gardner Duct Cyst (Mesonephric Cyst)
- •5.1.2 Bartholin Gland Cyst
- •5.2.1 Vaginal Infections
- •5.2.1.1 Vulvar Infections
- •5.2.1.2 Vulvar Thrombophlebitis
- •5.3 Vulvar Trauma
- •5.4 Vaginal Fistula
- •5.5 Post-Radiation Changes
- •5.6 Benign Tumors
- •6.1 Vaginal Malignancies
- •6.1.1 Primary Vaginal Carcinoma
- •6.1.1.1 MRI Findings
- •6.1.1.2 Lymph Node Drainage
- •6.1.1.3 Recurrence and Complications
- •6.1.2 Non-squamous Cell Carcinomas of the Vagina
- •6.1.2.1 Adenocarcinoma
- •6.1.2.2 Melanoma
- •6.1.2.3 Sarcomas
- •6.1.2.4 Lymphoma
- •6.2 Vulvar Malignancies
- •6.2.1 Vulvar Carcinoma
- •6.2.2 Melanoma
- •6.2.3 Lymphoma
- •6.2.4 Aggressive Angiomyxoma of the Vulva
- •7 Vaginal Cuff Disease
- •7.1 MRI Findings
- •8 Foreign Bodies
- •References
- •Imaging of Lymph Nodes
- •1 Background
- •3 Technique
- •3.1.1 Intravenous Unspecific Contrast Agents
- •3.1.2 Intravenous Tissue-Specific Contrast Agents
- •References
- •1 Introduction
- •2.1.1 Imaging Findings
- •2.1.2 Differential Diagnosis
- •2.1.3 Value of Imaging
- •2.2 Pelvic Inflammatory
- •2.2.1 Imaging Findings
- •2.3 Hydropyosalpinx
- •2.3.1 Imaging Findings
- •2.3.2 Differential Diagnosis
- •2.4 Tubo-ovarian Abscess
- •2.4.1 Imaging Findings
- •2.4.2 Differential Diagnosis
- •2.4.3 Value of Imaging
- •2.5 Ovarian Torsion
- •2.5.1 Imaging Findings
- •2.5.2 Differential Diagnosis
- •2.5.3 Diagnostic Value
- •2.6 Ectopic Pregnancy
- •2.6.1 Imaging Findings
- •2.6.2 Differential Diagnosis
- •2.6.3 Value of Imaging
- •3.1 Pelvic Congestion Syndrome
- •3.1.1 Imaging Findings
- •3.1.2 Differential Diagnosis
- •3.1.3 Value of Imaging
- •3.2 Ovarian Vein Thrombosis
- •3.2.1 Imaging Findings
- •3.2.2 Differential Diagnosis
- •3.2.3 Value of Imaging
- •3.3 Appendicitis
- •3.3.1 Imaging Findings
- •3.3.2 Value of Imaging
- •3.4 Diverticulitis
- •3.4.1 Imaging Findings
- •3.4.2 Differential Diagnosis
- •3.4.3 Value of Imaging
- •3.5 Epiploic Appendagitis
- •3.5.1 Imaging Findings
- •3.5.2 Differential Diagnosis
- •3.5.3 Value of Imaging
- •3.6 Crohn’s Disease
- •3.6.1 Imaging Findings
- •3.6.2 Differential Diagnosis
- •3.6.3 Value of Imaging
- •3.7 Rectus Sheath Hematoma
- •3.7.1 Imaging Findings
- •3.7.2 Differential Diagnosis
- •3.7.3 Value of Imaging
- •References
- •MRI of the Pelvic Floor
- •1 Introduction
- •2 Imaging Techniques
- •3.1 Indications
- •3.2 Patient Preparation
- •3.3 Patient Instruction
- •3.4 Patient Positioning
- •3.5 Organ Opacification
- •3.6 Sequence Protocols
- •4 MR Image Analysis
- •4.1 Bony Pelvis
- •5 Typical Findings
- •5.1 Anterior Compartment
- •5.2 Middle Compartment
- •5.3 Posterior Compartment
- •5.4 Levator Ani Muscle
- •References
- •Evaluation of Infertility
- •1 Introduction
- •2 Imaging Techniques
- •2.1 Hysterosalpingography
- •2.1.1 Cycle Considerations
- •2.1.2 Technical Considerations
- •2.1.3 Side Effects and Complications
- •2.1.5 Pathological Findings
- •2.1.6 Limitations of HSG
- •2.2.1 Cycle Considerations
- •2.2.2 Technical Considerations
- •2.2.2.1 Normal and Abnormal Anatomy
- •2.2.3 Accuracy
- •2.2.4 Side Effects and Complications
- •2.2.5 Limitations of Sono-HSG
- •2.3 Magnetic Resonance Imaging
- •2.3.1 Indications
- •2.3.2 Technical Considerations
- •2.3.3 Limitations
- •3 Ovulatory Dysfunction
- •4 Pituitary Adenoma
- •5 Polycystic Ovarian Syndrome
- •7 Uterine Disorders
- •7.1 Müllerian Duct Anomalies
- •7.1.1 Class I: Hypoplasia or Agenesis
- •7.1.2 Class II: Unicornuate
- •7.1.3 Class III: Didelphys
- •7.1.4 Class IV: Bicornuate
- •7.1.5 Class V: Septate
- •7.1.6 Class VI: Arcuate
- •7.1.7 Class VII: Diethylstilbestrol Related
- •7.2 Adenomyosis
- •7.3 Leiomyoma
- •7.4 Endometriosis
- •References
- •MR Pelvimetry
- •1 Clinical Background
- •1.3.1 Diagnosis
- •1.3.2.1 Cephalopelvic Disproportion
- •1.3.4 Inadequate Progression of Labor due to Inefficient Contraction (“the Powers”)
- •2.2 Palpation of the Pelvis
- •3 MR Pelvimetry
- •3.2 MR Imaging Protocol
- •3.3 Image Analysis
- •3.4 Reference Values for MR Pelvimetry
- •5 Indications for Pelvimetry
- •References
- •MR Imaging of the Placenta
- •2 Imaging of the Placenta
- •3 MRI Protocol
- •4 Normal Appearance
- •4.1 Placenta Variants
- •5 Placenta Adhesive Disorders
- •6 Placenta Abruption
- •7 Solid Placental Masses
- •9 Future Directions
- •References
- •Erratum to: Endometrial Cancer
Cervical Cancer |
157 |
|
|
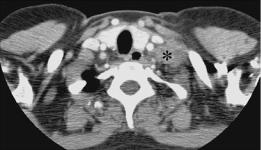
Fig. 35 Lymph node staging. Contrast-enhanced CT image. Supraclavicular lymph node metastasis on the left (asterisk). FIGO stage IVB
Dose-volume histograms serve to determine the respective dose corresponding to a specific organ volume such as the bladder or rectum, which are especially at risk. In planning the target volume to be irradiated (PTV, physical target volume), the primary tumor volume (GTV, gross tumor volume), the area of potential tumor extent (CTV, clinical target volume), and a safety margin that takes into account patient and organ motion are defined. The CTV comprises the uterus, at least the proximal third of the vagina, the parametrial tissue up to the pelvic wall, and the pelvic lymphatic drainage system, which is irradiated with a dose high enough to eliminate both micrometastases and manifest metastases. The only proven indication for para-aortic irradiation is metastatic para-aortic nodes. To spare the intestine, planning and irradiation are usually performed with the patient positioned prone on a belly board.
Modern radiation therapy for patients with cervical cancer is generally delivered with a combination of external beam radiation therapy (EBRT) and brachytherapy (BT), the latest being essential especially when therapy is administered with curative intent.
The advent of three-dimensional treatment planning of BT, allowing precise dose delivery to the target volume while limiting the dose to organs at risk, represents one of the most significant advancements in the treatment of cervical cancer. The introduction of computer tomog- raphy-based 3D treatment planning enables the
delineation the organs at risk as volumetric structures rather than arbitrary reference points, resulting in better estimation of maximum doses to the surrounding organs at risk as well as in improved local tumor control (Harkenrider et al. 2015). MR imaging is becoming increasingly used for image-based brachytherapy as it allows accurate demonstration of tumor size, location, and extension thanks to its superior soft-tissue resolution. In studies comparing MRI with CT-based treatment planning, MRI has shown to enable better tumor delineation and greater dose escalation (Harkenrider et al. 2015).
2.5\ Follow-Up
2.5.1\ Findings After Surgery
Scar tissue being older than 6 months has low signal intensity similar to that of muscle on T1and T2-weighted MR images. Fresh scars are of a higher signal intensity on T2-weighted images due to inflammation and neovascularization in the first months after surgery. Signal intensity decreases with fibrosis. This is why MRI should be performed not earlier than 6 months after the end of therapy and even then the signal intensities of recurrent tumor and scar tissue may still overlap on T2-weighted images. In these situations, a dynamic contrast-enhanced study could be necessary and help to better distinguish recurrent tumor on the basis of its earlier and more pronounced CM enhancement.
After radical hysterectomy, the uterus and the vaginal vault are absent (Brown et al. 1992). The vaginal stump is depicted with a smooth end and as a symmetrical, elongated, or rectangular structure that is sharply demarcated from the surrounding fatty tissue between the bladder and the rectum (Fig. 41). Cranial to the stump, the resection cavity is filled by the urinary bladder and bowel. Images in sagittal orientation are most suitable to evaluate the vaginal wall (Brown et al. 1992). On T2-weighted images, the vagina is characterized by a high-signal-intensity inner mucosal layer and a smooth outer muscular layer of low signal intensity. Surgical clips are depicted
158 |
F. Collettini and B. Hamm |
|
|
a |
b |
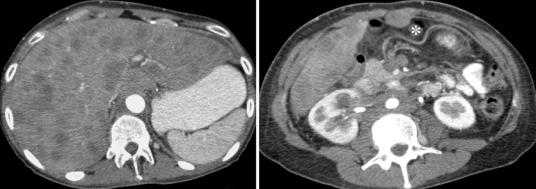
Fig. 36 Distant metastases. (a, b) Contrast-enhanced CT images. Numerous hypovascularized metastases are seen in the liver. In addition, metastatic spread to the perito-
neum (asterisk) and extensive para-aortic lymph node metastases. FIGO stage IVB
on MRI images as small signal voids at the vaginal stump. Some patients develop fibrotic scar tissue at the roof of the vaginal stump. It is characterized by an intermediate to low signal intensity on T2-weighted and T1-weighted images and may be difficult to differentiate from recurrence if it is of nodular configuration.
The periand postoperative complications include the development of vesicovaginal fistulas, which typically occur on the basis of necrosis roughly 1–2 weeks after the surgery. If they do not close spontaneously, they are operated on at 8 weeks or later in patients undergoing adjuvant radiotherapy. Injury to the urinary bladder occurs in 3–5% of patients undergoing radical hysterectomy and ureteral damage in 2%. Ureteral damage is usually treated by primary surgical repair. Ureterovaginal fistulas typically develop in the second postoperative week and have an incidence of about 1%. They spontaneously close after placement of a double-J catheter. Lymph edema of the legs has an incidence of 3% after surgery and of 5–15% after adjuvant radiotherapy. Clinically, approximately 15% of patients develop acute bladder voiding difficulties after radical hysterectomy with lymph node dissection, due to mechanical factors as well as damage to bladder innervation. The incidence increases with the radicalness of the operation.
In radical trachelectomy, MRI depicts the end- to-end anastomosis and the development of a posterior neofornix (Fig. 42). The latter may develop in the process of healing and must be carefully distinguished from a recurrent tumor. The few recurrences of cervical cancer observed after trachelectomy seem to occur at the site of anastomosis (Sahdev et al. 2005). Occasionally, stenosis has been observed after trachelectomy. Moreover, mobilization of the parametrial and paravaginal tissue can lead to diffuse thickening of the vaginal wall, which may mimic recurrent invasive tumor at MRI. Most of these postoperative changes recede spontaneously. In some patients, asymptomatic widening of the parametrial venous plexus has been observed. In patients becoming pregnant after trachelectomy, a cerclage is placed to keep the cervix closed.
Following lymph node dissection, metal clips are often seen at the pelvic wall as focal artifacts of low signal intensity at MRI or as metal-dense structures at CT. Lymphoceles most frequently develop after lymphadenectomy. They are usually small, cause no symptoms, and recede without therapy. If a lymphocele becomes symptomatic or infection is suspected due to contrast enhancement of the wall, therapy may be required in the form of repeat operation, puncture, drainage, or sclerotherapy.

Cervical Cancer |
159 |
|
|
Fig. 37 Distant metastases. T1w TSE image in sagittal orientation. Para-aortic lymph node metastases with vertebral infiltration of L1–L3. Consecutive total collapse of vertebral body of L2. FIGO stage IVB
Pelvic exenteration is the curative method of choice in patients with central pelvic tumor recurrence and comprises colpectomy and hysterectomy with removal of the bladder (anterior exenteration) or of the bladder and rectosigmoid (complete exenteration). In addition, the intervention may be performed as supralevator exenteration with partial resection of the levator plate or as translevator exenteration with vulvectomy and radical resection of the levator muscle, urogenital diaphragm, and vulvoperi-
neal soft tissue. The patient’s quality of life is improved by subsequent reconstruction of the pelvic organs with deep rectal anastomosis, creation of a urinary pouch, and possibly creation of a neovagina. The intervention-related mortality is about 5%.
2.5.2\ Findings After Chemotherapy
The main criterion for a response to chemotherapy is a reduction of tumor size. For follow-up of tumor size under chemotherapy, the largest transverse diameter of the lesion is measured according to the RECIST criteria (Eisenhauer et al. 2009). A size reduction of at least 30% is defined as partial response to therapy. Tumor progression is assumed when there is an increase in size of at least 20%. MRI is the method of choice for evaluating the response to chemotherapy in patients with cervical cancer. However, in patients on chemotherapy, both vital tumor tissue and inflammatory reactive areas are present, which are of high signal intensity on T2-weighted images and show early contrast enhancement on T1-weighted images. This is why reliable evaluation of tumor tissue on the basis of signal intensity at T2-weighted imaging and contrast enhancement at T1-weighted imaging is not always possible at this stage. Therefore, the most important criterion for a response to therapy at this stage is the size reduction of the tumor. The concomitant reaction of the surrounding tissue and fibrosis impairs not only the radiologic diagnosis but also later surgical treatment. In recent years, diffu- sion-weighted imaging (DWI) has been investigated as an alternative imaging modality for following up tumor response to chemotherapy. The idea behind the use of DWI to monitor tumor response relies on the fact that as the tumor shrinks with treatment, water mobility increases. Thus, the apparent diffusion coefficient (ADC) may increase and may serve as an early indicator of tumor response. Preliminary studies using DWI-MRI as an early biomarker to assess tumor response to neoadjuvant chemotherapy have been promising, raising the need for future large-scale prospective studies (Fu et al. 2012).
160 |
F. Collettini and B. Hamm |
|
|
a |
b |
c |
d |
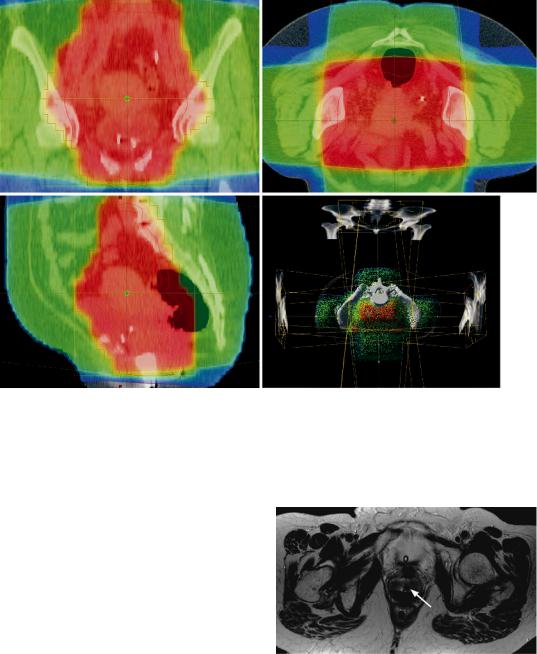
Fig. 38 Planning CT scan prior to radiotherapy. (a–c) Planning of irradiation therapy with determination of the target volume using the transverse planes of the CT scan and transfer to the sagittal and coronal planes. (d) Dose target volume and arrangement of fields using a four-field
technique. The target volume comprises the vagina, uterus, and locoregional lymphatic drainage system including a safety margin. Irradiation in the prone position to spare the small intestine (Courtesy of Dr. L. Moser, Berlin)
2.5.3\ Findings After Radiotherapy
MRI is the first-line radiologic modality for fol- low-up after radiation therapy. It may be performed 6 and 12 months after completion of irradiation and whenever tumor recurrence is suggested by the clinical or gynecologic findings. During and shortly after radiotherapy, the entire irradiated field shows a reactive signal increase on T2-weighted images and more pronounced contrast enhancement on T1-weighted images. This is why differentiation of tumor tissue from reactively inflamed tissue is impaired during and shortly after irradiation. Hence, MRI during or within the first 6 months after radiotherapy is indicated in exceptional cases for the
Fig. 39 MR imaging during intracavitary brachytherapy. Axial T2w image during intracavitary brachytherapy facilitates treatment planning and is useful in controlling the relationships between tumor and applicator (arrow). Measuring probes are placed in the rectum (courtesy of Prof. Dr. S. Marnitz, Köln)

Cervical Cancer |
161 |
|
|
a |
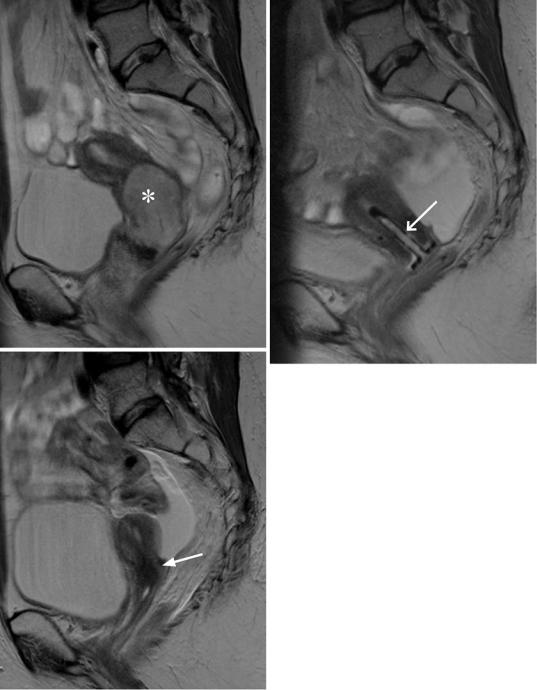
b |
Fig. 40 Status after hysterectomy. (a, b) T2w TSE images of different patients in sagittal orientation. Normal appearance of the vaginal stump (arrows) after hysterec-
tomy without (a) and with (b) filling of the vagina. Patient is catheterized
evaluation of tumor shrinkage or assessment of a fistula.
Effective radiation therapy leads to a significant reduction or complete disappearance of the tumor and a decrease in signal intensity on T2-weighted images (Figs. 43 and 44). A reliable sign of complete tumor remission is the return of the normal anatomy of the cervix and proximal vagina, which is suggested by the depiction of a homogeneous stroma of low signal intensity with a smooth mucosal layer often accompanied by shrinkage of the cervix. Recurrent tumor is typically seen as a high-signal-intensity mass corresponding to the original tumor on T2-weighted images.
The signal changes seen at MRI correlate with the overall radiation dose applied. Radiation-induced edema persists in the myometrium of the uterine corpus for up to 6 months after irradiation. After completion of radiation, there is a decrease in signal intensity on T2-weighted images, the endometrium becomes narrower, and the zonal anatomy of the myome-
Fig. 41 Status after trachelectomy. A T2w TSE image in sagittal orientation. Markedly shortened uterine cervix after radical trachelectomy with fertility-sparing uterovaginal reanastomosis (arrow). Gel filling of the vagina
trium is eliminated. In postmenopausal women, the uterus returns to its former MRI appearance without zonal anatomy after resolution of radiation -induced edema. The vagina also has an increased signal intensity on T2-weighted
162 |
F. Collettini and B. Hamm |
|
|
a |
b |
c
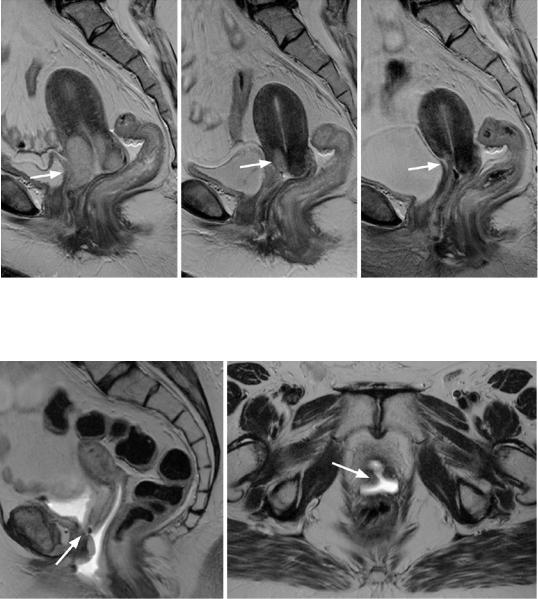
Fig. 42 Monitoring of radiotherapy, (a–c) T2w TSE images in sagittal orientation, (a) cervical cancer (asterisk) with infiltration of the vagina and parametria. (b) Tumorous mass of the cervix has disappeared 2 months
after radiotherapy. Endocervical sheath in place (arrow). (c) Normal appearance of the cervix and atrophy of the uterus 12 months after completion of irradiation (arrow). Small amounts of free fluid
Cervical Cancer |
163 |
|
|
images due to edematous and inflammatory changes in the acute and subacute phase after irradiation. About 6 months after the end of radiation therapy, there is a fibrosis-related signal reduction on T2-weighted images. Shrinkage of the cervix and vagina may occasionally lead to an effective stenosis (radiogenic fibrosis)
with subsequent development of hydrometra or hematometra. This condition is associated with symmetric enlargement of the uterus with a central fluid collection, which is of high signal intensity on T2-weighted images and also on T1-weighted images if the protein or blood content is high.
a |
b |
c |
Fig. 43 Monitoring of radiotherapy. (a–c) T2w TSE images in sagittal orientation. (a) Cervical cancer (arrow) with infiltration of the urinary bladder. (b) Size reduction
of the tumor (arrow) during radiotherapy. (c) No circumscribed tumor of the cervix is depicted 3 months after completion of irradiation (arrow)
a |
b |
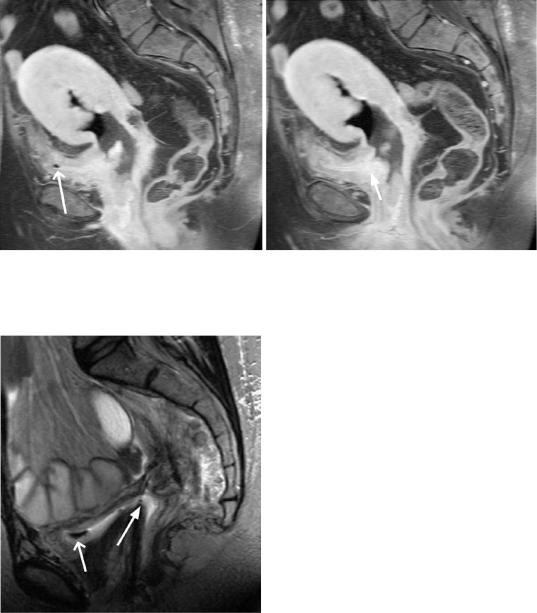
Fig.44 Fistula after radiochemotherapy. (a, b) T2w TSE images in sagittal and transverse orientation. Following radiochemotherapy of advanced cervical cancer, a fistula
depicted as a high-signal-intensity fluid-filled connection is seen between the vagina and urinary bladder (arrows). There is urine in the vagina
164 |
F. Collettini and B. Hamm |
|
|
a |
b |
Fig. 45 Fistula after radiochemotherapy. (a, b) T2w TSE images in sagittal and transverse orientation. Following radiochemotherapy of advanced cervical cancer, a fistula
depicted as a high-signal-intensity fluid-filled connection is seen between the vagina and urinary bladder (arrow). There is air in the bladder (open arrow)
Fig. 46 Fistula after hysterectomy. A T2w TSE image in sagittal orientation. Following hysterectomy for cervical cancer, a fistula depicted as a high-signal-intensity fluidfilled connection is seen between the vagina and urinary bladder (arrow). There is air in the bladder (open arrow)
A complication after irradiation is the development of fistulas due to therapy-induced regression of invasive cervical cancer (Figs. 45, 46, and 47). Contrast-enhanced T1-weighted images
identify a fistula as abnormal enhancement surrounding the low-signal-intensity fistular canal. A fistula from the uterus or vagina to the bladder is suggested when there is air in the bladder (Kim and Han 1997).
Postactinic radiogenic colitis is characterized by concentric edematous thickening of the intestinal wall with preservation of the layered structure and may be associated with additional edematous thickening and infiltration of the perirectal fat. Radiation-induced stricture of the ureter or insufficiency fracture of the sacral bone has become rare. The bone marrow of the pelvic bone in the irradiated field is regularly replaced with fat marrow, which is depicted on T1-weighted images with a high signal intensity.
In recent years, several authors have investigated the potentials of functional MRI including diffusionand perfusion-weighted imaging in evaluating the therapeutic response and predicting the clinical outcome after radiation therapy alone or in combination with chemotherapy. DCE-MRI, that involves intravenous application of low-molecular-weight paramagnetic intravascular contrast agents, enables the noninvasive assessment of tumor perfusion and oxygenation, major factors in response of tumor cells
